< img src =" https://images.obesityhelp.com/articles/wp-content/uploads/2016/10/AspireAssist.png" > October 26, 2016 The FDA just recently authorized a brand-new non-surgical, low risk, reversible procedure for patient regulated weight loss. The brand-new treatment is called AspireAssist.
The AspireAssist System
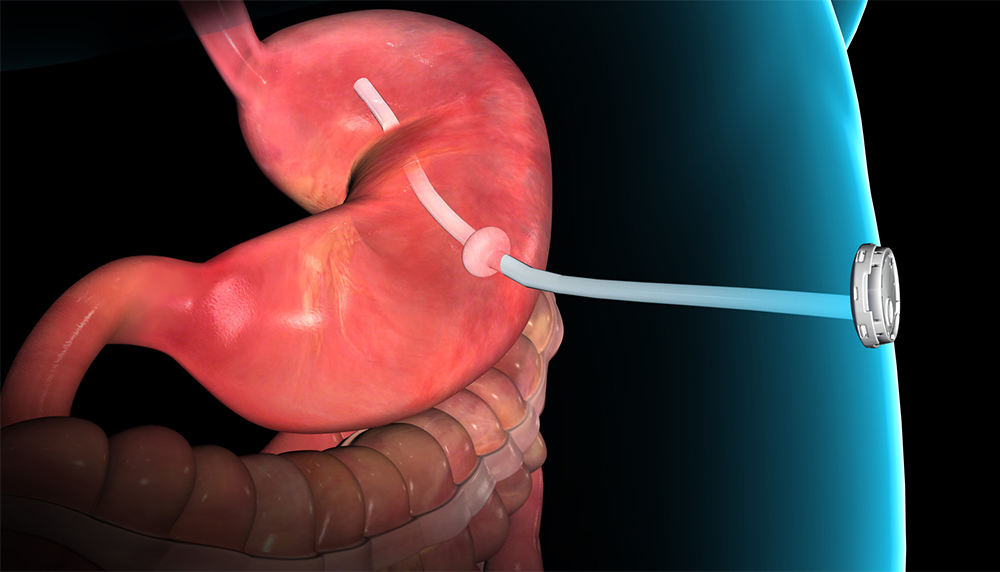
AspireAssist is an endoluminal weight loss device that causes weight-loss in overweight patients (BMI of 35-55). The gadget accomplishes weight reduction by 2 systems: reduction of calorie absorption and decrease of consuming rate. Over the first hour after eating a meal, the stomach secretes acid and starts to break down the food. Food consumed then enters the intestines where calories are taken in. The gadget makes it possible for patients to remove approximately 30% of stomach contents after a meal in the privacy of a toilet. Eliminating this volume lowers the number of calories a patient absorbs.
The inserted tube is just 6 mm in diameter. In order for stomach contents to drain pipes, the client should chew food extensively to fit through the tube. This requirement increases the typical meal usage time by more the 2 times standard. Slowing down the eating process allows patients to achieve satiety with a lower food volume.
What You Need To Learn About AspireAssist
The AspireAssist is inserted under sedation in an outpatient setting. Gadget insertion requires no special preparation on the part of the patient and is accomplished in about 15 minutes. Clients recuperate quickly after the treatment and normally go home in 2 hours after treatment completion. A lot of clients are back at work within a few days.
Outcomes: The AspireAssist was assessed in a research study performed at 10 leading organizations throughout the US. The trial enrolled 171 patients who were followed for 52 weeks. Clients who got AspireAssist lost 14.9% of body weight at one year compared to 4.9% for control patients- which implies AspireAssist patients lose 3 times as much weight as patients trying to utilize a diet plan. European research studies have revealed that clients lose 19% of body weight in the first year and reach a weight loss of 23.5% of body weight by the end of two years.
Security: The AspireAssist has a very low complication rate. Many patients tolerate the treatment well with just moderate discomfort at the insertion website. Most common side effects are experiencing pain at the insertion site, nausea/vomiting associated to sedation utilized for anesthesia, granulation tissue establishing at the insertion website, and peristomal irritation. All side effects are easily managed with workplace treatments or medication.
Tracking: Considering that the AspireAssist drains gastric contents including acid and electrolytes, it is essential that clients be kept track of regularly for metabolic and electrolyte balance. Clients must also go back to keep an eye on weight loss progress and stoma site health.
Satisfaction: Patient satisfaction is really high. 93% of patients are really pleased with AspireAssist and its weight loss and 96% of clients would refer a pal for the procedure.
With AspireAssist, patients manage the rapidity of their weight reduction. No unique diet plan is required however success requires patients to make gradual way of life modifications and modify they way they eat food.
Many patients in the United States continue to deal with obesity. For lots of patients, their insurance does not cover weight-loss surgery. For other patients, the dangers of surgical treatment avoid them from taking that route. AspireAssist is a reversible, non-surgical, low-risk treatment that can assist patients lose weight and maintain that weight-loss long term.
Dr. Shawn Garber and the team at New York Bariatric Group are the first to have actually positioned the first AspireAssist device in the United States because FDA approval. Their group is looked for to train other weight-loss cosmetic surgeons around the nation, and to assist develop new techniques and bariatric surgery choices, such as the AspireAssist.
 |
< img src=" https://images.obesityhelp.com/articles/wp-content/uploads/2016/10/angst.png "alt=" angst” width=” 120″ height= “160”/ > ABOUT THE AUTHOR Dr. John Angstadt is a board certified and fellowship qualified surgeon with vast experience in general surgery, transplant surgical treatment, in addition to minimally intrusive and bariatric surgery He established and developed the only bariatric surgery program in South Georgia. Dr. Angstadt has knowledge in numerous weight-loss surgeries and is also the director of the comprehensive and multidisciplinary medical weight-loss program at New York Bariatric Group.Read more posts from Dr. Angstadt! |
 |
< img src="https://images.obesityhelp.com/articles/wp-content/uploads/2016/10/chow.png" alt="chow" width="120" height="160"/ > ABOUT THE AUTHOR Dr. Jeffrey Chiao is a board licensed and fellowship qualified cosmetic surgeon with broad experience in minimally intrusive and bariatric surgical treatment. He has carried out hundreds of bariatric surgical treatments and treatments. At the New York City Bariatric Group, Dr. Chiao has actually been involved in the pioneering and cutting edge bariatric treatments, consisting of the Intragastric balloon and AspireAssist treatments. |
The FDA just recently approved a treatment for patient controlled weight loss The treatment is AspireAssist. Find out more about it from New York City Bariatric Group!.







